What Every Lawn Nutrient Actually Does (And What Happens When They’re Wrong)

You got your soil test back. The numbers are all over the page. Phosphorus is high, potassium is low, pH is 5.8, calcium is borderline, iron says "adequate" but your grass is still yellow. What does any of this actually mean for your lawn? The lab report tells you what you have. It does not tell you what it does. This guide fills that gap.
Every nutrient in your soil has a specific function in your grass. When one is deficient, you get visible symptoms (yellowing, browning, thin turf). When one is excessive, it often blocks another nutrient from being absorbed, creating a deficiency in something that is technically present in the soil. pH controls availability of nearly everything. On Long Island's sandy soils (CEC 3 to 8), nutrients leach faster than on clay or loam, making regular soil testing and targeted correction critical.
pH: The Master Switch
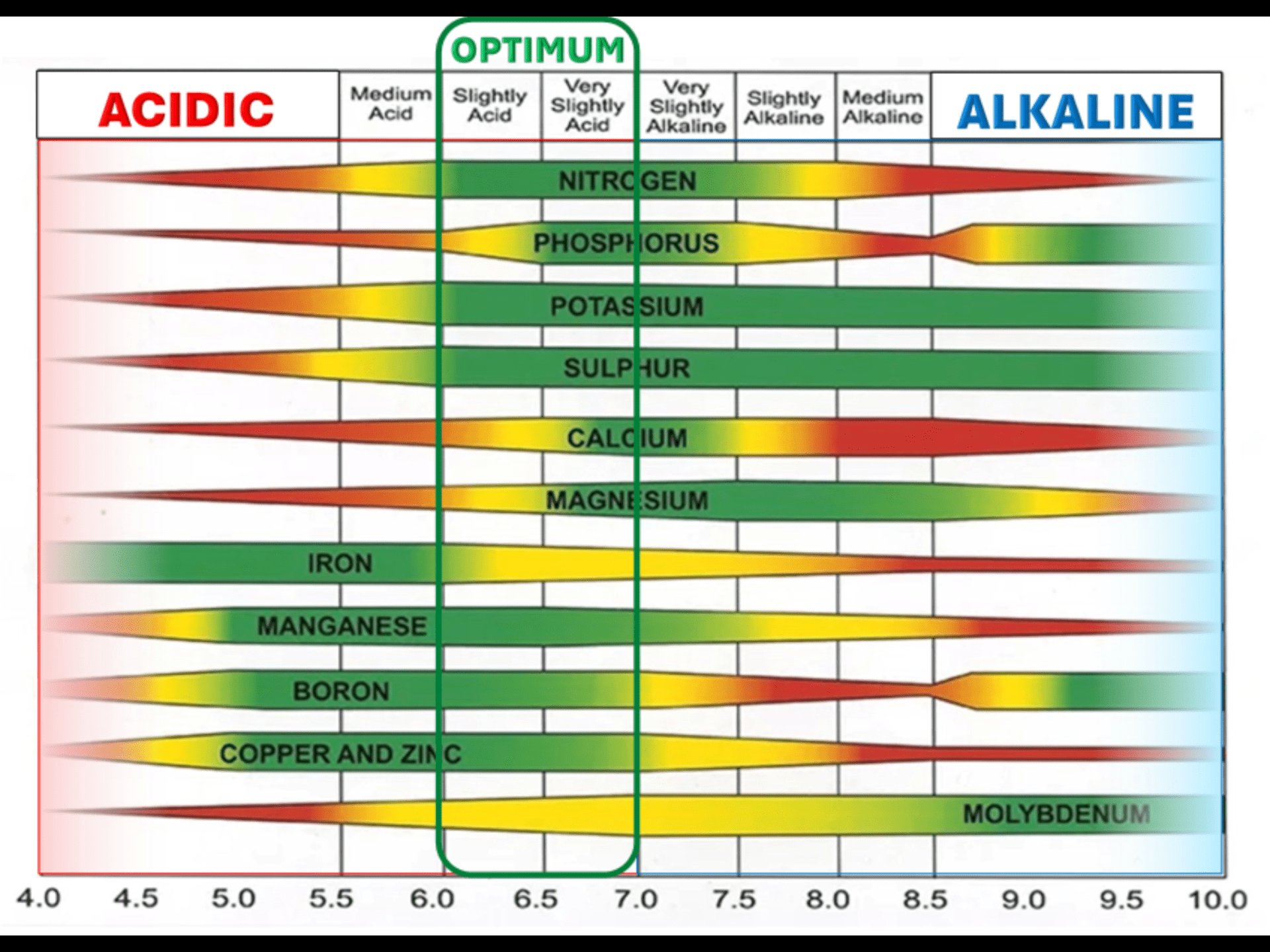
pH is not a nutrient. It is the control system that determines whether every other nutrient can reach your grass. Get pH wrong and nothing else on this page matters, because your lawn physically cannot access the nutrients you're paying for.
Cool-season grasses thrive between pH 6.0 and 7.0. In that window, every major and minor nutrient is at or near maximum availability. Move outside that range in either direction and things start locking up.
| pH Range | What Happens | What You See |
|---|---|---|
| Below 5.0 | Aluminum and manganese become toxic. Calcium and magnesium unavailable. Beneficial microbes die. | Stunted growth, thin turf, grass fails to respond to fertilizer. |
| 5.0 to 5.5 | Phosphorus gets locked by aluminum/iron compounds. Nutrient uptake drops. | Slow growth, purplish tinge on leaves, weak root system. |
| 5.5 to 6.0 | Slightly acidic. Most nutrients available but not optimal. | Lawn looks decent but not its best. Responds well to lime. |
| 6.0 to 7.0 | The sweet spot. Maximum availability of all nutrients. | Deep green, thick turf, strong root system, good disease resistance. |
| 7.0 to 7.5 | Iron, manganese, and zinc start locking out. | Interveinal yellowing on new growth (iron chlorosis). Common on LI. |
| Above 7.5 | Severe micronutrient lockout. Phosphorus locks up again. | Persistent yellowing that won't respond to fertilizer. Needs sulfur. |
Homeowner sees yellow grass. Throws down more nitrogen. Grass stays yellow. Throws down more. Still yellow. The problem was never nitrogen. It was pH above 7.0 locking out iron. A $5 soil test from Cornell Cooperative Extension would have caught this in 2 weeks. Instead, excess nitrogen leached straight into the aquifer.
Our complete pH guide covers how to test, how to correct with lime or sulfur, and the exact rates for Long Island soils.
The Big Three: Nitrogen, Phosphorus, and Potassium
Nitrogen (N): The Gas Pedal
Nitrogen is the nutrient most responsible for growth and color. It is the primary component of chlorophyll (the molecule that makes grass green) and the main driver of leaf production. When you fertilize your lawn, nitrogen is almost always the active ingredient doing the heavy lifting.
| Too Low | Just Right | Too High |
|---|---|---|
| Pale yellow-green color across the entire lawn | Rich, dark green color | Unnaturally dark blue-green color |
| Thin turf that won't fill in | Dense, thick growth with strong recovery | Excessive top growth (mow constantly) |
| Slow growth even in spring | Moderate, steady growth rate | Thatch buildup from rapid blade production |
| Older leaves yellow first (nitrogen is mobile) | Even color from old to new growth | Increased disease susceptibility (brown patch, pythium) |
| Weak root system | Balanced root-to-shoot ratio | Shallow roots (all energy goes to leaves) |
A moderate Long Island lawn needs about 2.75 lbs of nitrogen per 1,000 sq ft per year, split across 5 rounds. Our fertilizer calculator guide breaks down the exact math. On sandy soils, slow-release nitrogen sources are critical because quick-release N flushes through the root zone in days.
Phosphorus (P): The Root Builder
Phosphorus drives root development, energy transfer (ATP production), and seedling establishment. It matters most during renovation, overseeding, and for new lawns. In established lawns, phosphorus is often already adequate or high because it does not leach from soil the way nitrogen and potassium do. It binds to soil particles and stays put.
| Too Low (below 15 ppm) | Just Right (21 to 50 ppm) | Too High (above 50 ppm) |
|---|---|---|
| Purple/reddish discoloration on leaves | Strong, deep root system | Locks out iron, zinc, and manganese |
| Weak, shallow roots | Efficient energy production | Major environmental runoff concern |
| Poor establishment of new grass | Good stress and cold tolerance | Algae blooms in waterways |
| Slow fill-in after overseeding | Healthy seedling development | Banned in many states for lawn use |
Most established Long Island lawns test high for phosphorus because years of fertilizer applications have built up P levels that don't leach away. If your soil test shows P above 50 ppm, use a phosphorus-free fertilizer (look for the middle number being 0, like 24-0-11 or 32-0-4). Adding more phosphorus to already-high soil wastes money and creates the exact nutrient lockout problem described above.
Potassium (K): The Immune System
Potassium does not make your grass grow or turn green. What it does is keep your grass alive when things go wrong. It regulates water movement in cells, strengthens cell walls, activates disease-fighting enzymes, and builds cold hardiness. Think of it as your lawn's immune system and stress shield.
| Too Low (below 40 ppm) | Just Right (40 to 120 ppm) | Too High (above 120 ppm) |
|---|---|---|
| Brown, scorched leaf edges (marginal scorch) | Maximum disease resistance | Blocks magnesium and calcium uptake |
| Wilts easily in heat, slow to recover | Strong drought tolerance | Salt stress at extreme levels |
| Poor winter survival, winter kill | Good cold hardiness | Excess leaches (wasted money) |
| Gets every disease that comes through | Quick recovery from damage and traffic | Antagonizes other cation nutrients |
On Long Island's sandy soils, potassium is the nutrient that leaches fastest. CEC of 3 to 8 means the soil has almost no ability to hold onto K ions. You can apply potassium in April and it is gone by June. This is why fall potassium applications (winterizer) are critical: they build cold hardiness right when the plant needs it.
See Exactly Where Your Nutrients Stand
Upload your soil test results to the Blade Boss Soil Test Command Center. It maps every nutrient to optimal ranges, flags lockouts, and builds a correction plan for your specific soil.
The Secondary Nutrients: Calcium, Magnesium, and Sulfur
These three get overlooked because they are not on the front of the fertilizer bag. But they are just as important as N-P-K for Long Island lawns, and their interactions with each other cause some of the most confusing problems homeowners face.
Calcium (Ca): The Structural Engineer
Calcium builds cell walls, improves soil structure, and helps roots penetrate deep. It should occupy 60 to 70% of your soil's base saturation (the percentage of CEC sites holding calcium vs other nutrients). Lime is the primary source, which is why lime does double duty: it raises pH AND adds calcium.
Low calcium means weak cell walls (grass tears instead of cutting clean), poor root growth, and soil that compacts easily. High calcium raises pH and can block potassium and magnesium uptake.
Magnesium (Mg): The Chlorophyll Core
Magnesium literally sits at the center of every chlorophyll molecule. Without enough of it, your lawn cannot photosynthesize efficiently. The classic symptom is interveinal chlorosis on OLDER leaves (the veins stay green but the tissue between them turns yellow). This is different from iron chlorosis, which shows on NEW leaves.
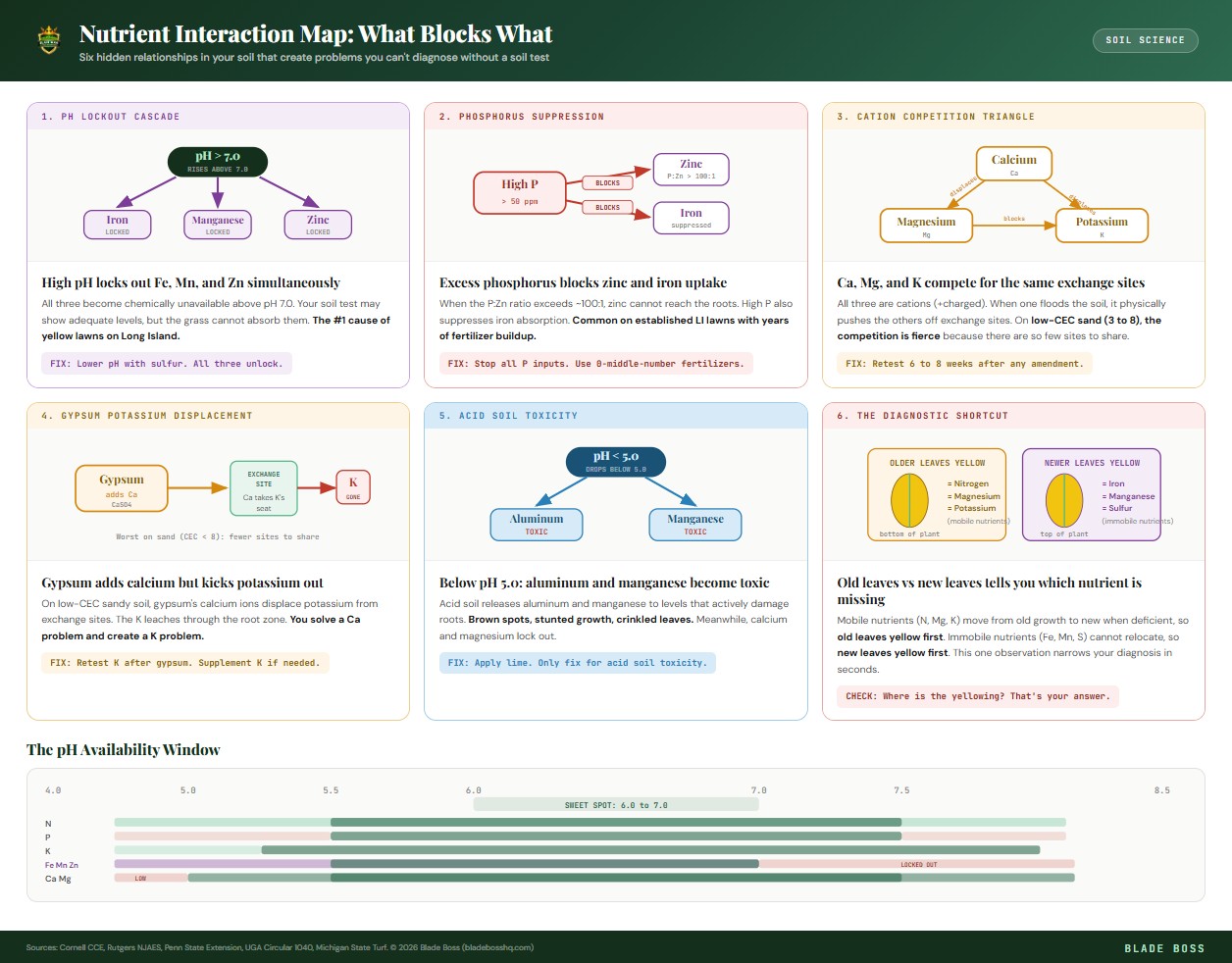
Yellowing on older, lower leaves first = magnesium deficiency (or nitrogen). Yellowing on newer, upper leaves first = iron deficiency (or manganese). This one observation can save you from chasing the wrong problem for an entire season. Magnesium is mobile in the plant (moves from old to new growth when short), iron is immobile (new growth suffers first).
Sulfur (S): The Protein Builder
Sulfur is essential for amino acids, proteins, and enzymes. Deficiency looks almost identical to nitrogen deficiency (uniform yellowing) with one critical difference: sulfur symptoms show on NEW growth first, while nitrogen symptoms show on OLD growth first. Sulfur is increasingly deficient in modern soils because industrial pollution (which ironically deposited sulfur via acid rain) has been cleaned up.
Elemental sulfur is also the primary tool for lowering soil pH when your soil is too alkaline. Our pH guide covers application rates.
The Micronutrients: Small Amounts, Big Impact
Micronutrients are needed in tiny quantities (parts per million instead of pounds per thousand), but their absence causes dramatic symptoms. These are the nutrients that cause the most confusion because their deficiencies look similar to each other and are often caused by pH lockout rather than actual absence from the soil.
Iron (Fe): The Color King
Iron is responsible for chlorophyll synthesis and that deep, dark green color everyone chases. Iron deficiency is the single most common micronutrient problem on Long Island because our water tends to push soil pH toward the alkaline side, and iron availability drops sharply above pH 7.0.
The symptom is textbook: interveinal chlorosis on NEW leaves (yellow tissue between green veins). Older leaves stay green because iron is immobile in the plant. The fix is almost never "add more iron." The fix is lower your pH so the iron already in your soil becomes available. Chelated iron (EDDHA form) works as a short-term bandage while sulfur corrections take effect.
Manganese (Mn) and Zinc (Zn): Iron's Partners
Manganese and zinc follow the same pH lockout curve as iron. Above pH 7.0, all three become increasingly unavailable. Manganese deficiency shows as a spotted or patterned chlorosis (often confused with iron). Zinc deficiency causes stunted, narrow leaves and distorted growth.
The key zinc interaction: high phosphorus suppresses zinc uptake. If your soil test shows P above 75 ppm and Zn below 2 ppm, the phosphorus is the culprit. No amount of zinc supplementation will fix it until you stop adding P and let levels drop naturally over time.
Copper (Cu) and Boron (B): Handle with Care
Copper deficiency is rare in lawns but causes blue-green color and wilting despite adequate water. The bigger concern is copper toxicity: it is toxic at relatively low concentrations and persists in soil for years. Never apply copper products without a soil test confirming a deficiency.
Boron has the narrowest safe range of any nutrient. The gap between "deficient" and "toxic" is measured in fractions of a ppm. Boron toxicity causes leaf tip burn that progresses inward, and once excess boron is in the soil, it persists. Correct boron deficiency only under professional guidance.
One text when growing degree days reach your next fertilizer window. No spam, just timing.
No spam. One seasonal alert per application window. Unsubscribe anytime.
Nutrient Interactions: When One Problem Creates Another
This is where soil chemistry gets interesting and where most homeowners get lost. Nutrients do not operate independently. They interact, compete, and block each other. Understanding these interactions is the difference between fixing a problem in one season and chasing your tail for three.
The pH Lockout Cascade
pH does not affect one nutrient at a time. When pH rises above 7.0, iron, manganese, AND zinc all lock out together. You get stacked deficiency symptoms that look like a disaster but have a single root cause. Fix the pH and all three come back online without supplementing any of them.
The same works in reverse. Below pH 5.0, aluminum and manganese become toxic (too available) while phosphorus, calcium, and magnesium lock out. One pH correction (lime) resolves all five problems simultaneously.
High Phosphorus Suppresses Zinc
When the P:Zn ratio exceeds roughly 100:1 in the soil, phosphorus actively suppresses zinc uptake at the root level. Your soil test can show zinc at 3 ppm (technically adequate) but the plant cannot absorb it because phosphorus is at 300+ ppm. This is why established lawns that have received years of high-P fertilizer sometimes develop zinc deficiency symptoms despite never having had a problem before.
The Calcium-Magnesium-Potassium Triangle
Calcium, magnesium, and potassium are all positively charged ions (cations) that compete for the same exchange sites on soil particles. When one is excessively high, it physically displaces the others from those sites and they leach away. This matters enormously on Long Island's low-CEC sandy soils because there are fewer exchange sites to go around.
- Excess calcium (from over-liming) displaces potassium and magnesium. Your K and Mg drop even though you never stopped applying them.
- Excess magnesium competes with calcium uptake and can cause soil compaction even on sandy soils. If your Ca:Mg ratio drops below 3:1, calcium supplementation may be needed.
- Excess potassium blocks magnesium absorption. This is why the K:Mg ratio matters: if potassium base saturation is more than double the magnesium percentage, Mg deficiency symptoms can appear.
- On sand (CEC 3 to 5), the competition is fiercer because there are so few exchange sites. One heavy lime application can strip your potassium levels for the rest of the season.
Calcium: 60 to 70% of CEC. Magnesium: 10 to 20%. Potassium: 2 to 5%. Sodium: below 3% (above 5% = soil structure damage, salt stress). Hydrogen: 5 to 15% (higher = more acidic). These are guidelines, not laws. If all individual nutrient ppm levels are above their critical minimums, the exact ratios are less important than the absolute amounts.
Gypsum and the Potassium Problem
Gypsum (calcium sulfate) is recommended when you need calcium without raising pH. But on low-CEC sandy soils (below CEC 8), gypsum introduces calcium ions that displace potassium from exchange sites. The potassium leaches and you end up solving a calcium problem while creating a potassium problem. On sandy Long Island soils, always retest 6 to 8 weeks after gypsum application and be prepared to supplement K.
CEC and Organic Matter: The Soil Foundation
CEC (cation exchange capacity) is not a nutrient. It is a measurement of how many nutrients your soil can hold. Think of it as the size of your soil's bank account. High CEC soil holds onto potassium, calcium, and magnesium between applications. Low CEC soil lets them flush through with every rain.
You cannot change your CEC overnight. It is largely determined by soil texture (sand vs clay) and organic matter content. The one thing you CAN do is increase organic matter through compost topdressing, which slowly raises CEC over time. Every 1% increase in organic matter adds roughly 2 meq/100g to your CEC. That is a meaningful improvement on a sandy soil starting at CEC 5.
Our sandy soil guide covers the full strategy for building CEC on Long Island soils, and the soil test interpretation guide walks through reading your CEC number and what it means for your fertilizer program.
The Diagnostic Cheat Sheet
When your lawn shows symptoms, start here:
| Symptom | Most Likely Cause | Check First | Common Misdiagnosis |
|---|---|---|---|
| Yellow lawn (entire blade, uniform) | Nitrogen deficiency | N rate and timing | Iron deficiency |
| Yellow between veins, NEW leaves | Iron lockout (pH too high) | Soil pH | Nitrogen deficiency |
| Yellow between veins, OLD leaves | Magnesium deficiency | Mg ppm and Ca:Mg ratio | Iron deficiency |
| Purple/red tinge on leaves | Phosphorus deficiency | P ppm (rare on LI) | Cold stress |
| Brown, scorched leaf edges | Potassium deficiency | K ppm | Drought stress or salt burn |
| Stunted, narrow leaves | Zinc deficiency | Zn ppm AND P levels | Herbicide damage |
| Brown spots, crinkled leaves | Manganese toxicity (pH too low) | Soil pH (below 5.5?) | Disease (fungal) |
| Leaf tip burn, margins yellow | Boron toxicity | B ppm (very narrow range) | Drought scorch |
| Uniform yellow on NEW growth only | Sulfur deficiency | S ppm | Nitrogen deficiency |
| Thin turf, won't fill in | Multiple possible (N, P, pH, compaction) | Full soil test | Just needs more seed |
If your lawn looks wrong and you don't know why, a soil test is always the right first step. A $5 test from Cornell CCE or a MySoil home kit tells you exactly what is high, what is low, and what is locked out. Stop guessing. The data costs less than a single bag of fertilizer.
What every number on your soil test means, the optimal ranges for Long Island soils, and exactly what to do when something is off. Two pages, print and keep with your test results.
Why This Matters More on Long Island
Long Island sits on a sole-source aquifer. Every nutrient that leaches past your root zone enters our drinking water supply. There is no backup. Excess nitrogen causes nitrate contamination. Excess phosphorus causes algae blooms in our bays and harbors. Over-applying because you did not understand your soil test is not just a lawn problem. It is an environmental problem.
And on our sandy soils, the margin for error is thinner than anywhere else. Low CEC means nutrients flush fast. One heavy rain after a heavy application and your money is in the water table. The entire point of understanding what each nutrient does is so you can apply exactly what your lawn needs, in the amount it needs, at the time it needs it. Nothing more.
See what Blade Boss members get and get access to the Soil Test Command Center, the fertilizer calculator, and zone-specific timing alerts that take the guesswork out of every application.
See Plans →Chris is a Combat Search and Rescue pilot turned airline pilot who built Blade Boss to bring military-grade precision to backyard lawn care. He learned nutrient interactions the hard way: by over-liming his Ronkonkoma lawn and watching potassium crash. Now the soil test comes first.
Related Reads
The soil test interpretation guide walks through reading your lab report step by step. The pH correction guide covers lime and sulfur application rates. The fertilizer schedule maps out all 5 rounds for the year. And the sandy soil guide explains why Long Island lawns need a different approach than what the bag says.
Frequently Asked Questions
Why is my lawn yellow even though I fertilize regularly?
Yellowing despite fertilization usually means one of three things: your soil pH is too high and is locking out iron (interveinal chlorosis on new growth), your nitrogen is being applied but not absorbed because of a pH or CEC issue, or you have a magnesium deficiency (yellowing between the veins on older leaves). A soil test is the only way to know which one. On Long Island, iron lockout from high pH is the most common cause.
What does high phosphorus do to a lawn?
High phosphorus is rarely harmful to the grass itself, but it locks out iron, zinc, and manganese. This means your lawn can show micronutrient deficiency symptoms (chlorosis, stunted growth) even though those nutrients are present in the soil. High phosphorus is also a major environmental concern because it causes algae blooms when it runs off into waterways. Many states ban phosphorus in lawn fertilizers for this reason.
What is nutrient lockout in lawn soil?
Nutrient lockout is when a nutrient is physically present in the soil but chemically unavailable to the grass. The most common cause is pH. Above pH 7.0, iron, manganese, and zinc become increasingly locked out. Below pH 5.5, phosphorus gets locked up by aluminum and iron compounds. Lockout also happens when one nutrient is so high it blocks absorption of another, like high phosphorus suppressing zinc uptake or excess potassium blocking magnesium.
What is the ideal calcium to magnesium ratio for lawns?
The traditional target is a Ca:Mg ratio between 3:1 and 8:1, with most extension programs suggesting around 5:1 to 7:1 as ideal. However, modern turf science (from Penn State, Michigan State, and others) emphasizes that absolute sufficiency levels matter more than exact ratios. If both calcium and magnesium are above their critical minimums (calcium above 500 ppm, magnesium above 50 ppm), the ratio is less important than hitting individual targets.
Does sandy soil on Long Island affect nutrient levels?
Yes, significantly. Long Island sandy soils have low CEC (cation exchange capacity) of 3 to 8, which means the soil cannot hold onto positively charged nutrients like potassium, calcium, and magnesium. They leach through the root zone with every rain or irrigation cycle. This is why Long Island lawns need more frequent, lighter fertilizer applications and why soil test results often show low K, Ca, and Mg even after amendments. The soil is not broken. It just cannot hold nutrients the way clay or loam can.
Level Up Your Lawn Game
20+ pro calculators, satellite lawn mapping, and USDA zone-specific schedules. Built for Long Island and Northeast homeowners.




Create a free account to join the conversation.